Private equity firms can breathe a sigh of relief after a federal judge dismissed claims that threatened to establish a precedent for holding private equity firms liable for certain actions by their portfolio companies.

Reviewing, analyzing, and navigating compliance, enforcement, investigation, and litigation developments and trends in the state and federal regulatory landscape

Private equity firms can breathe a sigh of relief after a federal judge dismissed claims that threatened to establish a precedent for holding private equity firms liable for certain actions by their portfolio companies.

Introduction
The medicinal and recreational use and popularity of marijuana has continuously grown throughout the U.S. since Oregon became the first state to decriminalize marijuana in 1973. To date, thirty-eight states and the District of Columbia have implemented medical marijuana programs, twenty-five of which have also expanded into adult-use.

On May 17, 2024, Colorado Governor Jared Polis signed into law Senate Bill 24-205, the Colorado Artificial Intelligence (AI) Act, making Colorado the first U.S. state to enact comprehensive legislation regulating the use and development of AI systems. The act is designed to regulate the private-sector use of AI systems, particularly addressing the risk of algorithmic discrimination arising from the use of so-called “high-risk AI systems.” The law will take effect on February 1, 2026, and the Colorado attorney general (AG) has exclusive enforcement authority.

Across states, lawmakers often embrace “medical” marijuana as a precursor to establishing a “recreational” marijuana market. Although marijuana remains illegal at the federal level — and would remain illegal even under the proposed rescheduling — federal regulators also recognize the therapeutic potential of certain marijuana-adjacent substances. At the federal level, the U.S. Food and Drug Administration (FDA) has approved one drug product derived from the cannabis sativa L. plant and three synthetic cannabis-related drug products. At the same time, 38 states and Washington, D.C., have established legal frameworks for access to medical marijuana. Comparing these frameworks to FDA’s drug approval process reveals fundamental differences between state and federal approaches to determining whether cannabis is “therapeutic.”

Only one day after reports surfaced that the Drug Enforcement Administration (DEA) will proceed with rescheduling cannabis from Schedule I to Schedule III of the Controlled Substances Act (CSA), Senators Charles Schumer (D-NY), Cory Booker (D-NJ), and Ron Wyden (D-OR) reintroduced the Cannabis Administration and Opportunity Act (CAOA or the Act), a nearly 300-page bill that would create a framework for the comprehensive regulation and taxation of cannabis in the United States. Then, on May 16th, the Department of Justice issued its notice of proposed rulemaking to reschedule cannabis to Schedule III. Administrative and legislative approaches to cannabis reform each have their own strengths and weakness that must be carefully considered. In addition, these competing approaches offer an opportunity to highlight the political differences between administrative and legislative policy reform at the federal level.

North Carolina Attorney General Josh Stein and the U.S. Attorney’s Office for the Middle District of North Carolina have reached a $500,000 settlement with Sharon Raynes Halliday and RAPHA Healthcare Services LLC, resolving a false claims lawsuit filed in July 2022 related to requests for payment submitted to the North Carolina Medicaid program.

In the latest episode of Regulatory Oversight, Troutman Pepper attorneys Gene Fishel and Joel Lutz welcome guests Aurelia Lewis and Beth Saunders of Lewis Media Partners to discuss evolving privacy laws and their impact on small to mid-sized businesses. Aurelia, as founder and president, and Beth, as vice-president, highlight their wealth of knowledge and experience guiding companies through effective advertising including media channel planning and buying.
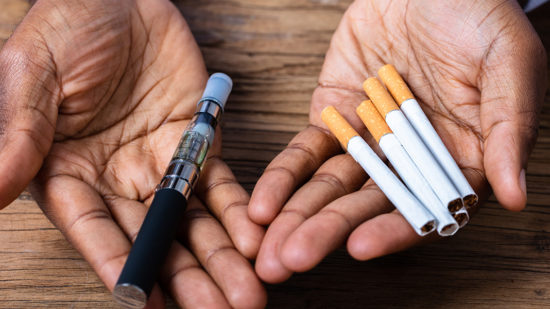
The landscape of tobacco product and cannabis flavor bans or restrictions varies significantly across the country. In both industries, some states restrict all or some flavors in all types of products, while other states restrict all or some flavors in some, but not all, products. Below, we provide a high-level overview of the flavor ban and restriction landscape in both industries. As we will discuss, there is a wide disparity between cannabis and tobacco product flavor bans or restrictions and, where they exist, there appears to be more flexibility among cannabis flavor restrictions than for tobacco product flavor bans or restrictions.

Published in Law360 on May 23, 2024. © Copyright 2024, Portfolio Media, Inc., publisher of Law360. Reprinted here with permission.
On April 23, the FTC promulgated its final rule[1] banning noncompetes nationwide.[2]
While most commentary has focused on the scrutiny noncompetes would now garner at the federal level, few discussed the waterfall enforcement effect at the state level that would follow as state attorneys general could deploy their broad authority under state unfair or deceptive acts or practices, or UDAP, laws to treat noncompetes as separate and independent violations.[3]

Last week the office of the Attorney General of Connecticut announced that the state had reached a settlement with HighBazaar over allegations that the organization allowed the unlicensed sale of cannabis, and the presence of minors, at their outdoor social cannabis events in Connecticut. The settlement represents one of many enforcement actions aimed at eliminating the state’s gray market and protecting licensed businesses and consumers.
In addition to cookies that are necessary for website operation, this website uses cookies and other tracking tools for various purposes, including to provide enhanced functionality and measure website performance. To learn more about our information practices, please visit our Global Privacy Notice.